A genetic mutation makes a glioma subtype resistant to radiation, but a new study finds a potential way around it using currently available drugs.
2:03 PM
Author |
Researchers from the University of Michigan Rogel Cancer Center have found that a genetic mutation seen in about half of all brain tumors produces a response that prevents radiation treatment from working. Altering that response using FDA-approved drugs restores tumors' sensitivity to radiation therapy, extending survival in mice.
The paper, representing more than five years of research, is published in Science Translational Medicine.
"These findings have great potential to impact medical treatment of patients with low-grade glioma, which is critically needed for this terrible disease," says senior author Maria G. Castro, Ph.D., R. C. Schneider Collegiate Professor of Neurosurgery and a professor of cell and developmental biology at Michigan Medicine.
The IDH1 gene is mutated in about half of gliomas, the most common type of brain cancer. These tumors tend to be low-grade and occur at a younger age. Survival can be seven to 10 years.
"The challenge is that following initial treatment with surgery and chemotherapy, the tumors always come back, and they come back with a vengeance," Castro says. "If we can intervene early in this diagnosis, before the tumor comes back more aggressive and deadly, it could have a big impact for these patients."
LISTEN UP: Add the new Michigan Medicine News Break to your Alexa-enabled device, or subscribe to our daily audio updates on iTunes, Google Play and Stitcher.
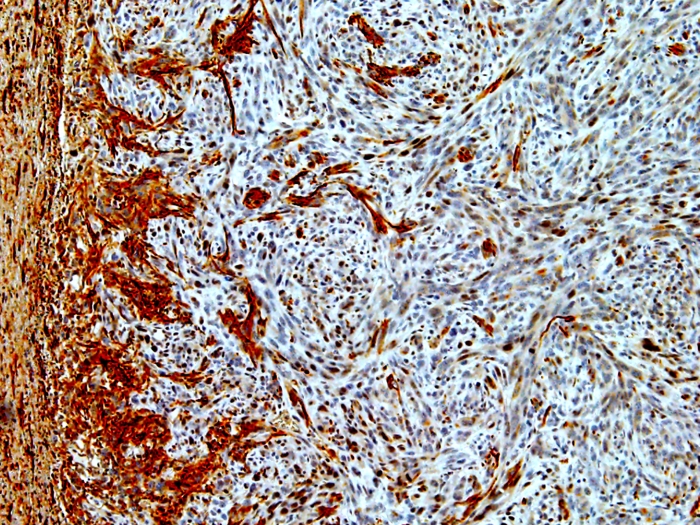
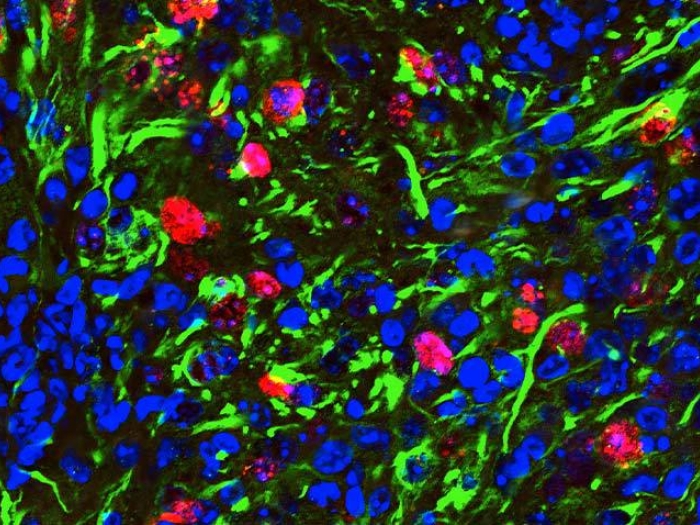
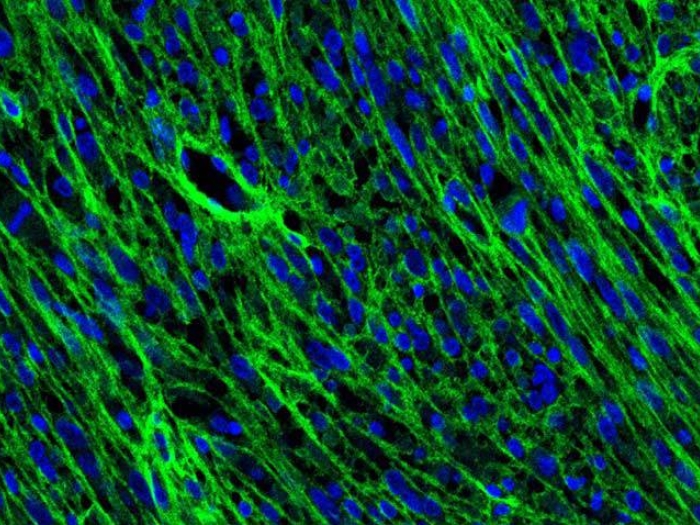
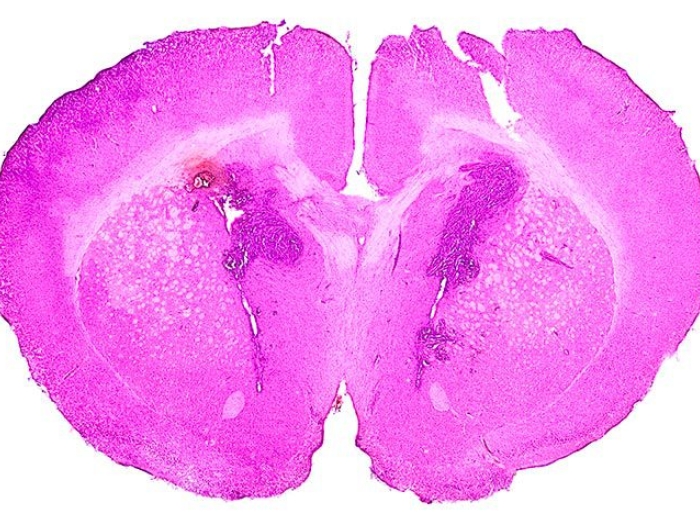
The researchers created a mouse model of IDH1-mutant glioma. The IDH1 mutation does not in itself cause cancer. For a tumor to form, mutations must occur in IDH1, p53 and ATRX.
The researchers discovered that when all three mutations are present, mutated IDH1 increases the tumor cells' DNA damage response. This reduces the efficacy of the radiation treatment, as the cancer cells can now correct the radiation-induced DNA damage.
If we can intervene early in this diagnosis, before the tumor comes back more aggressive and deadly, it could have a big impact for these patients.- Maria G. Castro, Ph.D.
Adding a drug that inhibits a critical pathway in DNA damage repair made the tumors sensitive to radiation treatment. Researchers initially discovered this using the newly-created mutant IDH1 mouse models and confirmed their findings in tissue samples from patients with IDH1-mutant glioma. This suggests that a precision medicine approach to gliomas would better direct their treatment.
"Our findings elucidate how mutant IDH1 interferes with current glioma treatments and provide a blueprint on how to overcome such interference. As a direct consequence of these data, we will implement a series of novel clinical trials at our institution in the near future," says Pedro R. Lowenstein, M.D., Ph.D., Richard C. Schneider Collegiate Professor of Neurosurgery and professor of cell and developmental biology.
MORE FROM MICHIGAN: Sign up for our weekly newsletter
The FDA has already approved DNA repair blockers to enhance sensitivity to chemotherapy or radiation in some cancers. The researchers are currently working with colleagues in radiation oncology, pathology, neuro-oncology and neurosurgery to translate their findings into a clinical trial to test the combination in patients with low-grade gliomas with IDH1 mutation.
Pathologists already test for IDH1 mutation as part of the diagnostic process because it influences prognosis and treatment, so this therapeutic strategy would be straightforward to implement in clinic.
"Now we have a potential new way of treating these tumors, that will make radiation deliver a much bigger impact," says lead study author Felipe J. Nunez, Ph.D., a research specialist in the Castro/Lowenstein lab. "By combining these two approaches, we hope that the new treatment will extend remission in these patients and potentially turn low-grade glioma into a chronic disease."

Explore a variety of health care news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!