In mice, researchers find the metabolite uridine can feed pancreatic cancer cells when glucose availability is low
11:00 AM
Author |
Researchers at the University of Michigan Rogel Cancer Center have discovered a new nutrient source that pancreatic cancer cells use to grow. The molecule, uridine, offers insight into both biochemical processes and possible therapeutic pathways.
The findings, published in Nature, show that cancer cells can adapt when they don’t have access to glucose. Researchers have previously identified other nutrients that serve as fuel sources for pancreatic cancer; this study adds uridine to the catalog.
Pancreatic tumors have few functioning blood vessels and can’t easily access nutrients that come from the bloodstream, like glucose. Costas Lyssiotis, Ph.D., Maisel Research Professor of Oncology and lead investigator of the study, explained that without the right nutrients, the cancer cells get hungry. “We know they still grow, obviously, but what are they using to grow?” he said. “These findings show that, under certain circumstances, uridine is one of those fuels.”
Asked about impact, Zeribe Nwosu, Ph.D., one of the co-first authors in the study, said “the ability of cancer to switch to alternative nutrients has fascinated me for a long time. Blocking such compensatory switches could lead us to new treatments and that’s the door we hope this study will open”.
Uridine is present in the tumor microenvironment, but its exact source, and how cancer cells access it, remains a mystery. “Part of the picture is it’s in the bloodstream, but we don’t know where it’s coming from specifically,” said Lyssiotis. “Likely, it’s coming from multiple places, and so far we haven’t been able to pin it to a single source.”
Events that Lyssiotis refers to as “times of crisis” – when cells don’t have enough nutrients, because of limited blood access and/or intense competition between cells – could be a clue as to why, and where, cells turn to uridine. "The cancer cells seem to be sensing the concentrations of glucose and uridine in the local environment to inform their adaptation,” said Matt Ward, another co-first author. Lyssiotis’ team recognize this unknown regulatory process, as well as a cancer-promoting mutation in the KRAS gene, which is common in pancreatic cancer, as two ways that cancer cells control their usage of uridine.
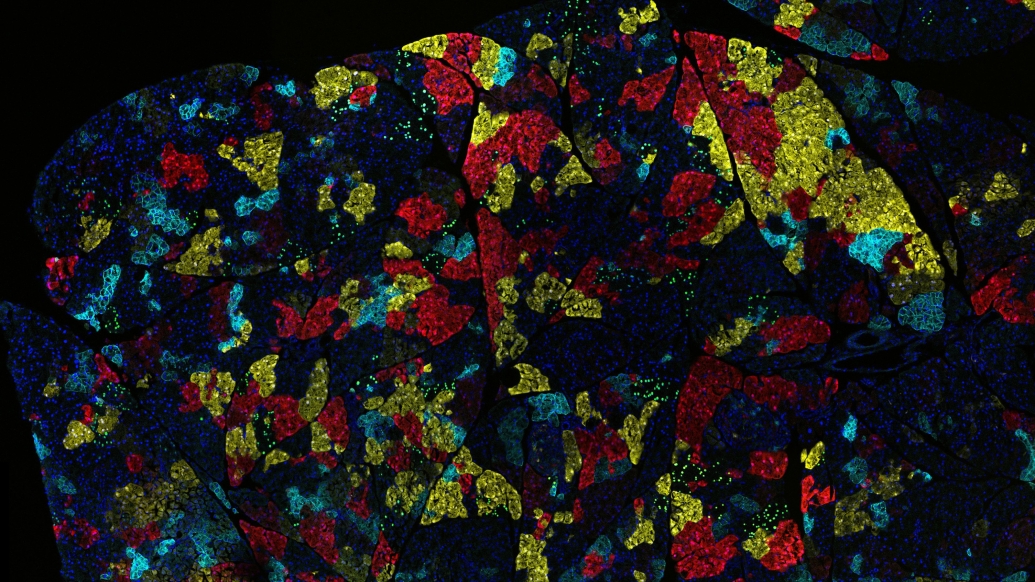
Lyssiotis and his team have been working on this research for nearly a decade alongside their collaborators in the Sadanandam lab at the Institute for Cancer Research in London. They used a technology that screens hundreds of different nutrients to see which ones support pancreatic cancer growth. Typically, researchers look at standard nutrients like sugar, protein and fat, but Lyssiotis’s team took an unbiased approach.
“We used a large panel with over 20 pancreatic cell lines and around 200 different nutrients to assess different ways pancreatic cancer cells grow,” he explained. “What do they actually metabolize? This method led us to discover uridine.”
This method offers therapeutic insight, too. The findings showed that uridine is metabolized by the enzyme uridine phoshorylase-1, or UPP1. Blocking UPP1 had a major impact on the growth of pancreatic tumors in mice, findings that suggest the importance of testing drugs that block uridine as possible new treatment options.
“There’s potential to better understand and treat pancreatic cancer with new drug targets and new therapeutic approaches,” said Sadanandam, co-author on the study.
More research is needed to determine the best way to move this discovery to the clinic.
Additional authors include Zeribe C. Nwosu, Matthew H. Ward, Peter Sajjakulnukit, Pawan Poudel, Chanthirika Ragulan, Steven Kasperek, Megan Radyk, Damien Sutton, Rosa E. Menjivar, Anthony Andren, Juan J. Apiz-Saab, Zachary Tolstyka, Kristee Brown, Ho-Joon Lee, Lindsey N. Dzierozynski, Xi He, Hari PS, Julia Ugras, Gift Nyamundanda, Li Zhang, Christopher J. Halbrook, Eileen S. Carpenter, Jiaqi Shi, Leah P. Shriver, Gary J. Patti, Alexander Muir, Marina Pasca di Magliano8 Anguraj Sadananda, Costas A. Lyssiotis.
COI: C.A.L. has received consulting fees from Astellas Pharmaceuticals, Odyssey Therapeutics, and T-Knife Therapeutics, and is an inventor on patents pertaining to Kras regulated metabolic pathways, redox control pathways in pancreatic cancer, and targeting the GOT1-pathway as a therapeutic approach (US Patent No: 2015126580-A1, 05/07/2015; US Patent No: 20190136238, 05/09/2019; International Patent No: WO2013177426-A2, 04/23/2015). A.S. received grants from Merck, Pierre Fabre and Bristol Myers Squibb and is an inventor on patents on Colorectal cancer classification with differential prognosis and personalized therapeutic responses (PCT/IB2013/060416); Prognostic and treatment response prediction in gastric cancer – Priority Patent CSC/BP7295892; Patient classification and prognostic method (GEP-NET) – Priority Patent – EP18425009.0; and Molecular predictors of therapeutic response to specific anti-cancer agents (US9506926B2).
Funding: Z.C.N. was supported by the Michigan Postdoctoral Pioneer Program at the University of Michigan Medical School, NIH/NCI grant K99CA267176 and NIH/NIGMS grant R25GM143298. M.H.W. by the CABRI Undergraduate Research Grant and NIH/NIGMS grant 5T32GM139774-02. P.S by NIH/NCI grants 5T32CA140044-12 and 5T32CA140044-13. M.R. by NIH/HD grant T32-HD007505. C.J.H by NIH/NCI grant K99/R00CA241357. E.S.C. by the Department of Veteran Affairs Career Development Award IK2BX005875. J.S. by NIH/NCI grants K08CA234222 and R37CA262209. A.M. by American Cancer Society (IRG-16-222-56), the University of Chicago Cancer Center Support Grant (P30 CA14599), the Pancreatic Cancer Action Network (2020 Career Development Award), the Brinson Foundation, the Cancer Research Foundation and the Ludwig Center for Metastasis Research. M.P. by NIH/NCI grants R01CA151588 and R01CA198074. A.S. was supported by two grants from Pancreatic Cancer UK, including Future Leaders Fund (FLF2015_05_ICR) and Research Innovation Fund (RIF2014_06_Sadanandam) and funding from Ian Harty Charitable Trust. C.A.L was supported by NIH/NCI grants R37CA237421, R01CA248160, and R01CA244931, and M.P and C.A.L. by UMCCC Core Grant P30CA046592. The funders had no role in study design, data collection and analysis, or the content and publication of this manuscript.
Paper cited: “Uridine-derived ribose fuels glucose-restricted pancreatic cancer,” Nature. DOI: 10.1038/s41586-023-06073-w

Explore a variety of health care news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!