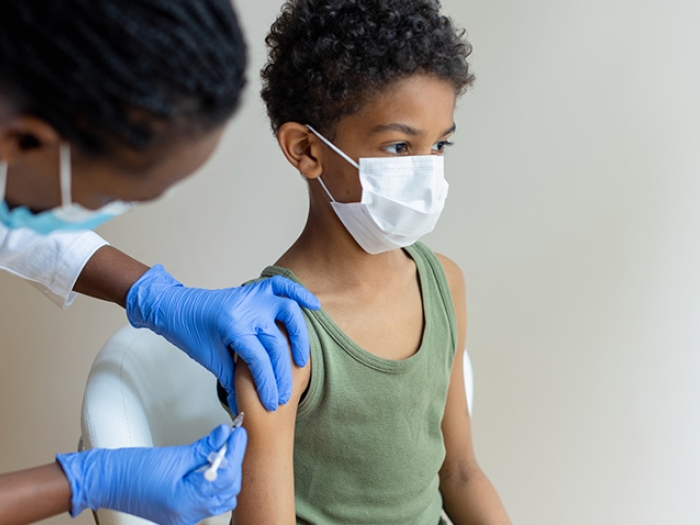
Kids 5-11 are now eligible for Pfizer’s COVID vaccine. Pediatric experts share answers to parents’ questions as they consider vaccinating their child.
10:59 AM
Author |
It's the pandemic milestone many families have been waiting for: a COVID-19 vaccine to protect younger kids.
Both the Federal Drug Administration and Centers for Disease Control and Prevention have officially approved emergency authorization for kids ages 5-11 to receive Pfizer-BioNTech's COVID-19 vaccine.
The approval paves the way for roughly 28 million more kids to be eligible for COVID-19 vaccination across the United States and comes at a pivotal moment with schools reopened and the highly contagious Delta variant affecting more children.
"This is a critical step to ensuring that all K-12 school-aged kids have the chance to be protected from COVID as the Delta variant spreads and we head into winter season," said Elizabeth Lloyd, M.D., a pediatric infectious diseases expert at University of Michigan Health C.S. Mott Children's Hospital.
It also comes after a peak of COVID-19 cases, hospitalizations and deaths among children in the U.S.
While kids' risk of severe illness from the virus is still low, there have been over 500 pediatric deaths related to COVID-19 and over 40 additional deaths due to a rare but serious COVID-linked condition called multi-system inflammatory syndrome (MIS-C), according to the CDC.
More than six million children have tested positive for COVID-19 since the beginning of the pandemic, with more than 1 million new cases reported in September 2021 alone. From late June to mid-August 2021, COVID-19 hospitalizations among children and adolescents increased fivefold.
But experts acknowledge that while many parents will be eagerly scheduling a vaccination for their child as soon as possible, others may have hesitations.
"We all want to make the right health decision for our children. It can be hard to filter through all of the information – and misinformation – that's been out there about COVID vaccines," Lloyd said.
Mott experts answer top questions below about the Pfizer COVID-19 vaccine for younger kids, including Lloyd and colleagues Christine Mikesell, M.D., a pediatric hospitalist who helped drive guidelines for treating children with MIS-C, and Jesse Hansen, M.D., a pediatric cardiologist who sees kids in the hospital's pediatric post-COVID-19 syndrome clinic.
What to know about the COVID-19 vaccine for kids under 12:
1. The FDA made its decision based on data from a clinical trial of the vaccine in children. What do those data show about the vaccine's impact on infections, symptoms and hospitalizations in younger kids?
Pfizer's clinical trial, which started in March, ultimately included roughly 4,500 children ages 5 to 11, with two thirds receiving a lower dose COVID-19 vaccine and others receiving a placebo in order to compare outcomes for efficacy and side effects.
The vaccine was 100% effective at protecting younger children from severe illness from COVID-19. The levels of antibody produced by children in this age group were also comparable to the immune response seen in kids 12 and over and adults.
As part of its review, the FDA did a risk-benefit analysis of the vaccine, particularly weighing the risk of a rare side effect causing a usually short-lived type of heart inflammation known as myocarditis, Lloyd said.
"The FDA and CDC determined that the vaccine produced a strong immune response in young children and found that in multiple scenarios, the benefits of the vaccine clearly outweighed the risks for this age group," Lloyd said.
2. Were the reactions and side effects seen in the kids in the clinical trial the same as what we've seen in adults and older kids?
Like older kids and adults, younger kids also mostly experienced minor side effects – more than two thirds had pain and soreness at the injection site, two fifths experienced fatigue and nearly a third reported headaches.
Side effects were worse after the second shot, but usually lasted just a couple of days.
Four serious adverse events occurred during the trial, but further investigations revealed that none of them were in any way related to the vaccine, Lloyd said.
3. Were there breakthrough cases in fully vaccinated kids in the clinical trial?
There were three cases of COVID-19 out of over 3,000 children who received the vaccine, but none of them led to severe illness.
4. Do young children also have a risk of myocarditis, which has been reported in teens receiving the Pfizer vaccine?
Myocarditis, a type of inflammation in and around the heart, is an extremely rare side effect of the COVID-19 vaccine that has predominately affected men under 30 and about 1 in 20,000 kids ages 12-17.
None of the children in the Pfizer trial for ages 5-11 experienced myocarditis but because it's so rare, experts will continue monitoring for it as a larger number of children receive the vaccine, Hansen says.
He notes that younger children are expected to have an even lower risk of the rare side effect because they're receiving a lower dose of the vaccine and are also less likely to get myocarditis in general due to their developing immune systems.
For people 12 and over who have experienced myocarditis after the second dose of the vaccine, symptoms are usually mild and short-lived, such as shortness of breath and chest pain that resolve after a couple of days.
"We're confident that this is an extremely rare event, but are going to continue watching it closely because our kids deserve it and parents trust us to remain vigilant," he said.
"Pediatric cardiologists, emergency medicine doctors and hospitalists across the country were able to quickly identify this really rare event in teens and young adults and collaborate with each other on how to best take care of those who experienced it," he added.
Notably, COVID-19 infections cause myocarditis at much higher rates than the COVID-19 vaccines do. Viruses are among the most common triggers of myocarditis, Mikesell says.
"For kids 12 and over, the risk of adverse side effects, including myocarditis, are much more likely to be caused by COVID itself than from the vaccine and are also more likely to be serious," she said.
5. What's the difference between the dose that FDA has authorized for this age group, and what teens and adults are already getting?
Kids ages 5-11 will receive about a third of the dose offered to those 12 and older – 10 micrograms compared to the 30 micrograms adults and teens receive.
The smaller dosing will also be distributed with smaller needles to make it easier for pediatricians and pharmacists to administer the vaccine to younger kids.
"This dose is what researchers determined to be the best to ensure a strong antibody response while minimizing side effects," Lloyd said.
There are currently studies to see if a lower dose may eventually be appropriate for adults as well, she says.
6. If a child is turning 12 soon or has a weight and height comparable to an older tween or teen, should they wait to get the bigger dose?
Doctors don't recommend waiting for a child to turn 12 in order to get a greater dose.
Vaccines don't work like medications because it's not a dose-response relationship, Lloyd explains. Bodyweight is more important for medication dosing because medications usually work after a certain level reaches the bloodstream.
For a vaccine to be effective, the cells of the immune system must be trained to recognize the component of the vaccine that mimics the virus. Later, these educated cells will recognize and be able to attack the real virus.
"A person who is taller or at a heavier weight may require more medication to see the same effects as someone who is smaller. But body size doesn't affect immune response in the same way, which is why some vaccines can even use the same dose for all ages," Lloyd said. "
"For vaccines to work, it's not about having a certain level of it in your blood, but rather stimulating the immune system. Children often only need only a fraction of the dose because they usually have more robust immune responses than adults."
The Pfizer vaccine for ages 5-11 was designed to be used in a wide spectrum of ages, says Mikesell who plans to get her own two young daughters vaccinated as soon as possible.
"We're recommending you go by your child's age and get the vaccine as soon as they're eligible," she said. "Even though the risk of severe COVID is lower in kids than adults, there's still a risk and it's better to be covered by vaccination longer rather than to wait."
7. What's the process for a child to be fully vaccinated?
Similar to kids over 12 and adults, children ages 5-11 will receive two doses of the vaccine three weeks apart. Children are considered fully vaccinated two weeks after their second dose.
8. If a parent is ready to get their child vaccinated, where will they be able to get it?
Once they're officially eligible, kids may be able to get a COVID-19 vaccine from their pediatrician, at retail pharmacies, health clinics, hospitals or even school-based sites depending on the location.
The White House has already expedited shipments of the vaccine to providers and community centers.
Unlike the vaccine rollout for adults, which prioritized high-risk groups, all kids ages 5 to 11 will be eligible to get the vaccine.
This is a critical step to ensuring that all K-12 school-aged kids have the chance to be protected from COVID-19.Elizabeth Lloyd, M.D.
Signing your child up for a COVID-19 vaccine could also be an opportunity to check in with their pediatrician and make sure they're up to date on all their other important shots, including the flu vaccine – which can be safely distributed at the same time as the COVID-19 vaccine – experts say.
9. Should kids only get the first shot and not the second to reduce side effect risks, which some other countries have recommended?
Hansen notes that because COVID-19 prevalence is much higher in the U.S. than other places that have followed this recommendation, the risk-benefit calculation for one dose isn't comparable.
"That may be a reasonable approach for other places but not the appropriate response to match the reality of what's happening here," he said.
Studies in adults also show that the vaccine doesn't offer the same protection after just one shot. One dose of the Pfizer vaccine was only 30% effective against the delta variant specifically.
"Both doses are necessary to get full immunity and we recommend full vaccination for all ages," Lloyd said.
10. Should children with allergies, underlying conditions or complex medical histories (such as those who have had chemo, blood clots, kidney disease, a heart transplant or congenital heart disease) hold off on a COVID-19 vaccination?
The Pfizer trial for ages 5-11 didn't exclude children with allergies, asthma or certain comorbidities, including obesity, congenital heart disease, cystic fibrosis and sickle cell disease among other conditions.
Just like with older kids, doctors don't recommend skipping vaccination because of an underlying condition. In fact, the opposite is true, experts say.
"Vaccination is particularly important for children who are immunocompromised in any way," Hansen said.
"We strongly recommend all kids get the COVID vaccine, including all of our heart patients, because it reduces the risk of both heart and non-heart related complications."
He notes that certain kids, such those who have recently had a heart transplant are at an even higher risk of infections.
"There may be some kids who are getting specific medications that may impact their immune response and make the vaccine less effective," he noted. "We recommend these families have conversations with their care teams as they plan for vaccinating their kids."
Those preparing for surgery, he notes, may also need to consult with doctors on the best timing for getting the vaccine.
The COVID-19 vaccine also doesn't contain any food or latex allergens. While allergic reactions to the vaccine are extremely rare, children with a history of any severe allergic reactions or anaphylaxis after a vaccine should consult with their provider as well.
11. If my elementary-aged child recently had COVID-19, how long should I wait to get them vaccinated?
Children may get vaccinated as soon as they feel better and no longer have symptoms, and are done with any required quarantine.
"We know kids get some natural immunity after a COVID infection, but we're not sure how long that lasts," Lloyd said. "We've also seen people get COVID multiple times unfortunately."
"It's definitely still beneficial for kids to get vaccinated after having COVID and I wouldn't recommend waiting longer than a couple of months."
12. If a child gets vaccinated, will they shed virus particles that could affect classmates or young siblings?
No, this isn't possible because there are no live viruses used in the COVID-19 vaccine.
While versions of weakened, non-harmful viruses have been used in some other vaccines, such as the measles, mumps and rubella vaccine, that is not the case for the mRNA COVID-19 vaccines used by both Pfizer and Moderna.
The mRNA creates a spike protein to help train the body how to identify and attack the real virus if it's encountered.
"No harmful viral particles are going to be shed from someone who got the COVID vaccine," Lloyd said.
13. If a child is generally afraid of needles, or has special needs such as autism, anxiety or a developmental condition, what can parents do to help them before and during the vaccination?
Needle anxiety is common but there are many steps you can take to prepare your kids. Using child-friendly language to explain why the shot is important to protect your child and those around them is helpful as well as using distractions like a video on an iPad or providing comfort positions for younger kids.
"Helping our child through a vaccination is all in how we set it up," Mikesell said. "Kids aren't usually worried about side effects like achiness or being tired later. Normally, it's the fear of that needle moment they've built up in their head.
"Parents should emphasize that the poke is just a few seconds long and that there will be people to help them."
Get more tips on preparing children for a COVID-19 vaccine from a Mott Child Life specialist.
14. If a child gets vaccinated do they still need to wear a mask in public, or in school?
Recommendations may vary depending on how prevalent COVID-19 is in the community.
Currently, both the American Academy of Pediatrics and CDC recommend masking in schools and indoors, but these guidelines may continue to evolve depending on whether infection rates go down and vaccination rates go up, Lloyd says.
15. Should parents be worried about long term side effects of the COVID-19 vaccine in kids?
History teaches us that not only are side effects from vaccines extremely rare, but biologically, there'd be no reason to see any emerge more than eight weeks after someone receives a vaccine.
Most side effects would appear within days or weeks, Lloyd says. It's always possible a very rare side effect is revealed only after tens of thousands of vaccines get administered but there's a tried and true process to monitor for these – even if it's a side effect that may only impact one in a million people.
Researchers now have data from nearly a year of COVID-19 vaccine administration in adults and it has been over seven months since kids ages 5-11 started being vaccinated in the clinical trials.
"We have decades of research and a rigorous vaccine development process to provide reassurance of vaccines' long-term safety," Lloyd said.
16. Will COVID-19 immunity wear off in kids over time like it has in adults, and if so will there need to be a COVID-19 booster or annual COVID-19 shot?
This question is still under active investigation, experts say.
"We don't have enough data to know if long term, we will need to take a flu-vaccine type of approach to COVID vaccines too," Hansen said.
"That may be determined by how well we work as a community to prevent the spread of the virus and mutations and if other variants show up."
17. What role do children play in spreading COVID-19 in their communities and vulnerable family members?
While severe COVID-19 sickness is less common in children, studies show that they are still able to transmit the virus to others.
"Children generally are still vectors of infection, even though they don't seem to be getting as sick as adults do," Lloyd said.
"They still have the potential to spread it to older people like grandparents or younger siblings."
"Not only does the vaccine benefit the child themselves but there's a secondary benefit of reducing their ability to spread COVID to others, especially if they will be around someone who is at higher risk of serious complications."
18. Do healthy kids even need to be vaccinated if their risk of severe COVID-19 is low?
While children are at a low risk of severe illness or death from COVID-19, some do get hospitalized and die.
Since the start of the pandemic, about 1.9 million children ages 5-11 have been infected, with more than 8,300 hospitalizations and 94 deaths, according to federal data.
The death toll related to COVID-19 puts the illness in the top 10 causes of death for kids over the last year.
Some kids are also experiencing other quality of life issues due to MIS-C and long-haul symptoms.
"Parents should keep in mind that while their child's risk of complications from COVID infection may be low, it's still higher than any risk associated to the vaccine," Hansen said. "Vaccination is a simple way to protect children from COVID risks, reduce community spread, and help prevent the development of new variants."
19. When will other COVID-19 vaccines be available to children under 12?
Moderna's vaccine for ages 6-11 is expected to be next in line to be reviewed by the FDA for emergency authorization. The company recently announced that its COVID-19 vaccine is also safe and effective and that it plans to submit data soon.
Johnson & Johnson's clinical trials for children as young as 12 are expected to start this fall.
20. What about kids younger than 5?
Data from the other two age cohorts from the Pfizer trial – children ages 2-5 and children 6 months to 2 years of age – are expected as soon as the fourth quarter of this year.

Explore a variety of healthcare news & stories by visiting the Health Lab home page for more articles.

Department of Communication at Michigan Medicine
Want top health & research news weekly? Sign up for Health Lab’s newsletters today!